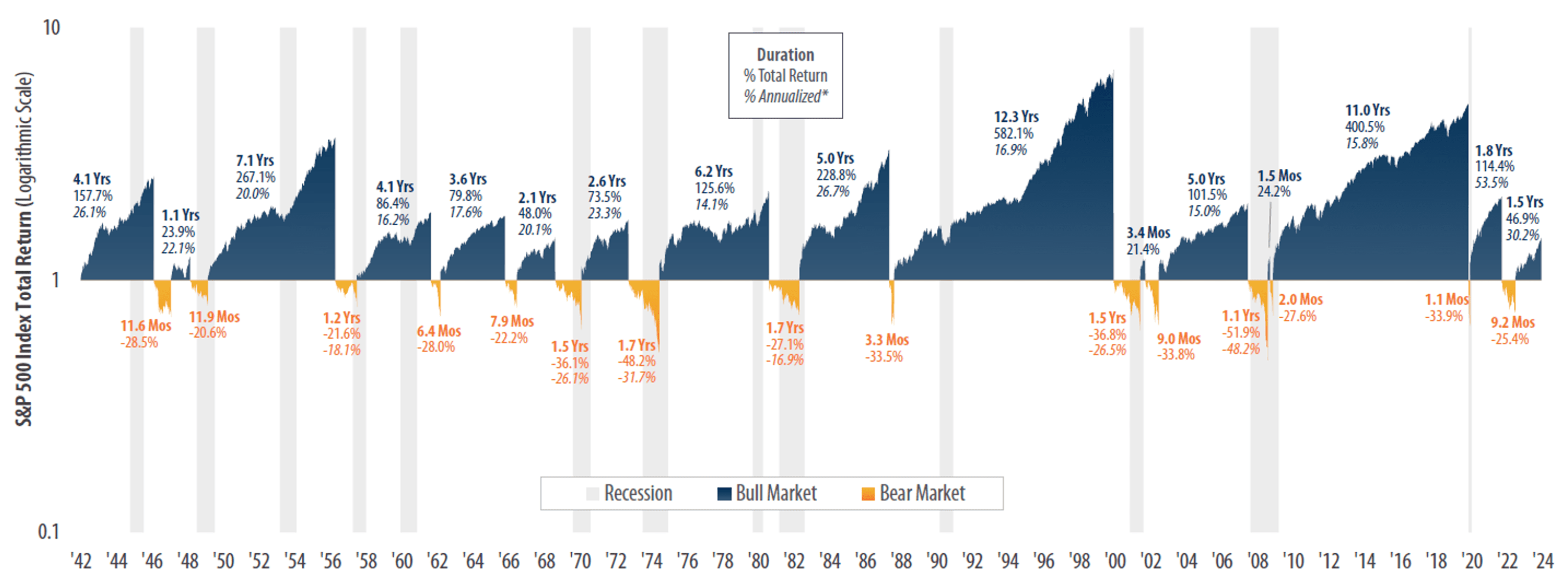
Note: This article was first published in The Business Times on 25 November 2020; data as of 19 November 2020 On 9 November 2020, Pfizer announced a wonderful development for mankind. Trial results from the pharmaceutical giant’s COVID-19 vaccine candidate, developed together with Bio NTech, showed that it could be 90% effective in preventing infection. A week later, Moderna revealed that its COVID-19 vaccine candidate was 94.5% effective in trials. This was followed by an update from Pfizer a few days later that its vaccine candidate was actually 95% effective . COVID-19 is still a serious global health threat. Pfizer and Moderna’s vaccines have yet to pass regulatory approvals at the time of writing (19 November 2020). Both companies have said too that they can supply their respective vaccines at scale only in 2021. Pfizer’s vaccine candidate also poses a significant logistical challenge since it needs to be transported...